Rechargeable Aqueous Li-air Batteries
Our objective is to develop rechargeable Lithium air batteries with energy densities far beyond those of Lithium ion batteries using safe, low cost materials and cost-efficient processing technologies. To realise battery systems that exceed current state of the art both in energy density and power density, we will therefore focus on Li-air batteries with aqueous catholytes with somewhat lower theore¬tical energy density, but a much higher potential to reach theoretical limits in practical devices. The envisaged aqueous Li-air battery chemistries are characterised by highly soluble discharge products so that key problems of their organic counterparts (clogging of the reactive cathode surface by Li salt deposits, limited power density & cycle efficiency, limited volumetric energy density by space reserved for salt deposition, sensitivity to water etc.) are automatically eliminated.
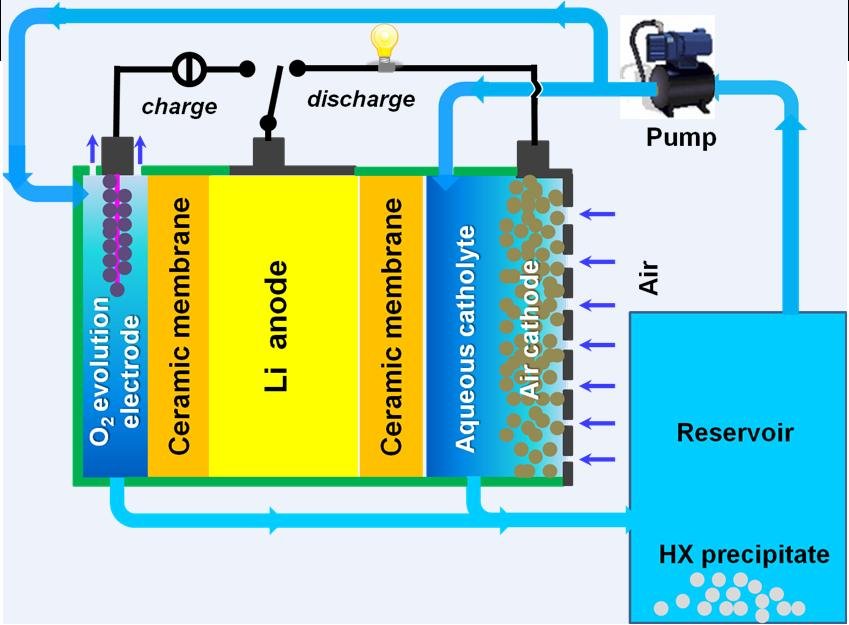
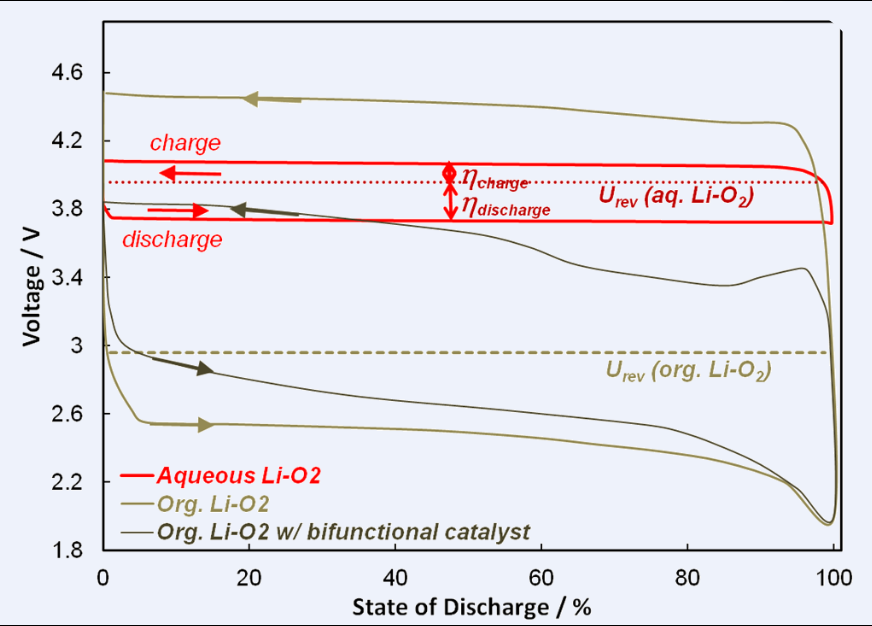
Our devices are designed using aqueous electrolytes that combine high solubilities for the discharge product with innovative protected anodes based on stable fast ion conducting solids (NASICON-type superionic conductors, garnet-related Lithium ion conductors and thio¬phosphates). We will incorporate novel designs that permit reversible operation beyond the solubility limit as well as maximize power performance, cycle efficiency, scalability and safety.
All-solid-state Batteries
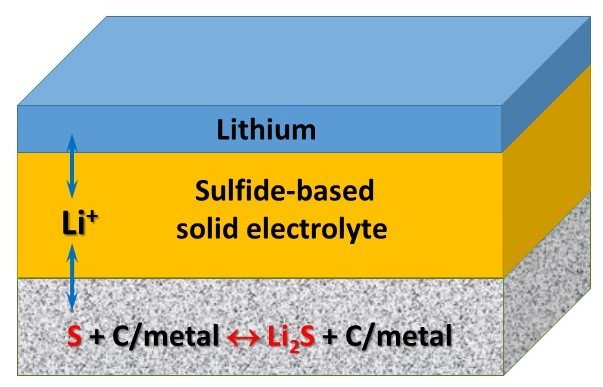
All-solid-state lithium secondary batteries, by replacing flammable liquid electrolyte with inorganic solid electrolyte, exhibit superior safety and reliability when compared to conventional lithium-ion batteries. Elemental sulfur is promising candidate of positive electrode materials due to its high theoretical capacity (1672 mAh/g), low toxicity and low cost. In combination with solid electrolyte, the resulting Li/S batteries are free from the issue caused by the dissolution of polysulfides in liquid electrolyte, leading to high cyclability. Sulfide-based fast Li+ ion conductors, such as lithium argyrodites, are employed as solid electrolytes, taking advantages of their high ionic conductivity. We design all-solid-state lithium secondary batteries using various sulfur composite cathodes such as sulfur/carbon composite and sulfur/metal composite and investigate their cyclic performance at different C-rates and temperatures.
Lithium-based All-solid-state Thin Film Microbatteries
The employment of solid electrolyte in a lithium ion battery (LiB) is generally considered safer than those of conventional liquid organic electrolyte by removing the issue of high flammability and high volatility. Ceramic solid electrolytes are believed to have potential advantages in terms of handling and chemical stability over liquid organic electrolyte. Therefore, all-solid-state LiBs and related all-solid state lithium batteries (ASSLBs) have attracted significant attention in recent years. Meanwhile, the increasing demand in microscale devices for various microsystem applications requires power sources of proportionate form factor. Lithium-based all-solid-state thin film microbatteries can become a promising contributor in this area while using fast Li+ ion conducting solids could extend the application range to higher power system.
New Fast Li+ and Na+ Ion Conducting Solids
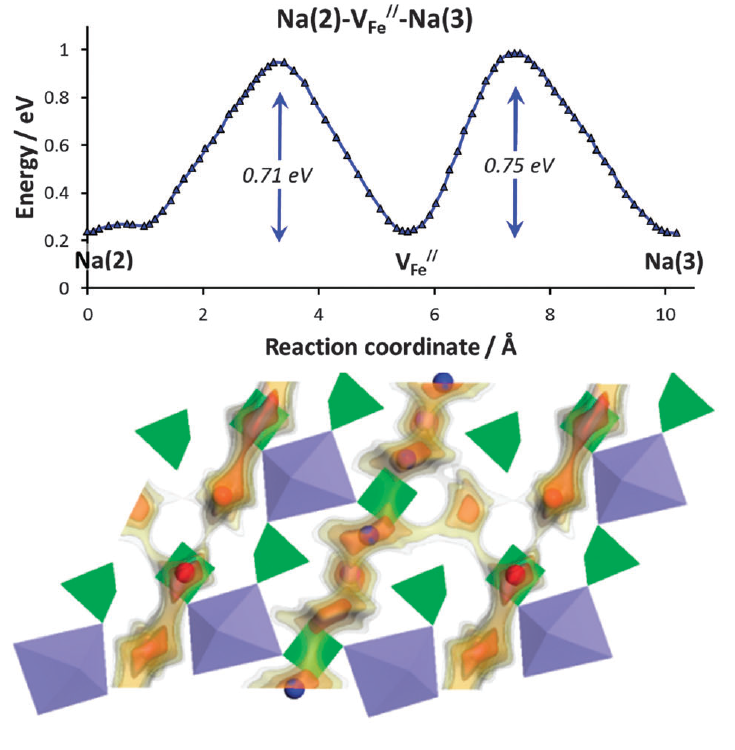
In this project, we aim to search for fast Na+ and Li+ ion conductors through computational screening. Based on the bond-valence based method using softBV parameters established by our own group,1 we are currently developing our software that extends from fast screening to efficient study of local structure models. Such tool allows us to determine the minimum energy pathways (MEP) involved in Na+ ion migration for the modelled crystal structure, such that a better understanding of the underlying contributions towards ionic conductivity can be obtained. An example is our recent work on the Na2+δFe2-δ/2(SO4)3 compound where the local structure models allow us to determine that cross-linking between fast 1D conducting channels due to iron deficiency could reconcile the reported good rate performance and the original crystal structure.2
2 L. L. Wong, H. M. Chen and S. Adams, Phys. Chem. Chem. Phys., 2015, 17, 9186-9193.
Bond-valence-based Modeling of Ion Transport (softBV forcefield)
The energy landscape of a mobile ion in a crystalline electrolyte structure can be generated with the softBV forcefield which is a Morse-type binary forcefield. The name comes from the fact that its parameters are deduced from the bond valence parameters for bonding between cation-anion. A coulomb repulsion term is added to account for the cation-cation interaction. We develop and maintain a software based on this forcefield and energy landscapes generated, and attempt to extend its use beyond visualization of conduction pathway, such as percolation analysis, vibration frequency analysis, dopant predictor and so on.
Organic Solar Cells
In the last decades, organic solar cell (OSC) has attracted substantial attention due to its advantages including low cost, light weight, mechanical flexibility and ease of process. Bulk heterojunction (BHJ) architecture based on blends of donor and acceptor polymers is usually used as the photoactive layer in OSC. The purpose of applying BHJ is to boost the dissociation of excitons and the transport of electrons and holes in opposite directions. Therefore, the performance of BHJ plays an important role in improving the overall efficiency of OSCs. In our group, we focus on various BHJ systems and donor-acceptor materials. Our main job is to look into the details of the morphology of these materials as well as their interfaces at atomic-level using different simulation methods, which is very difficult to be achieved experimentally.